RAMAN-LIBS publication released
The publication of our colleagues Virgina Merk, Wolfgang Werncke and Lutz Pfeifer was recently published in the “Analyst” of the Royal Society of Chemistry.
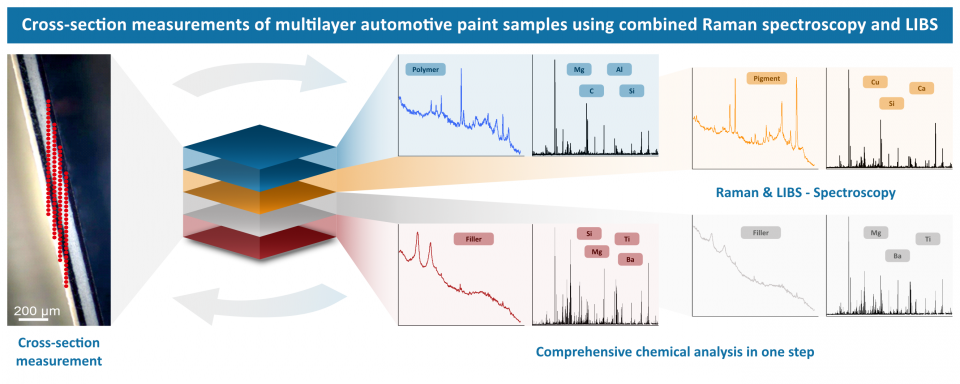
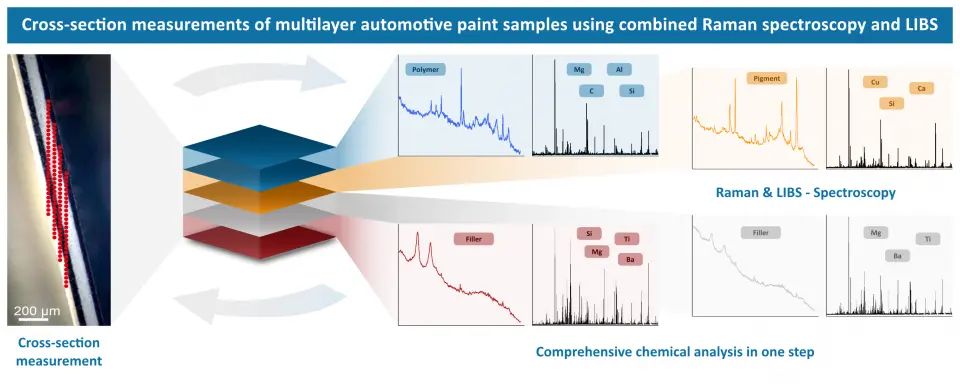
Our RAMAN-LIBS research group investigated the cross-sections of multilayer car paint samples. By using both techniques with our CORALIS system, LIBS with a nanosecond high energy pulse and Raman with a cw laser, both the elemental and molecular information and compositions become accessible. The ability to perform both measurements sequentially at the same measurement points provides a comprehensive picture of each layer. The approach can be a good additional option to established techniques for forensic analysis of car paint samples.
Full article: Analyst, 2022, DOI: 10.1039/D2AN01474B